 Infectious or pathogenic prions are resistant to proteases, because of their three-dimensional shape, which hides away parts of the prion that would normally fit in proteases and which would cause the prion to be digested. Proteases have clefts, or subpockets, into which proteins fit, where the substrate (protein) gets cut. 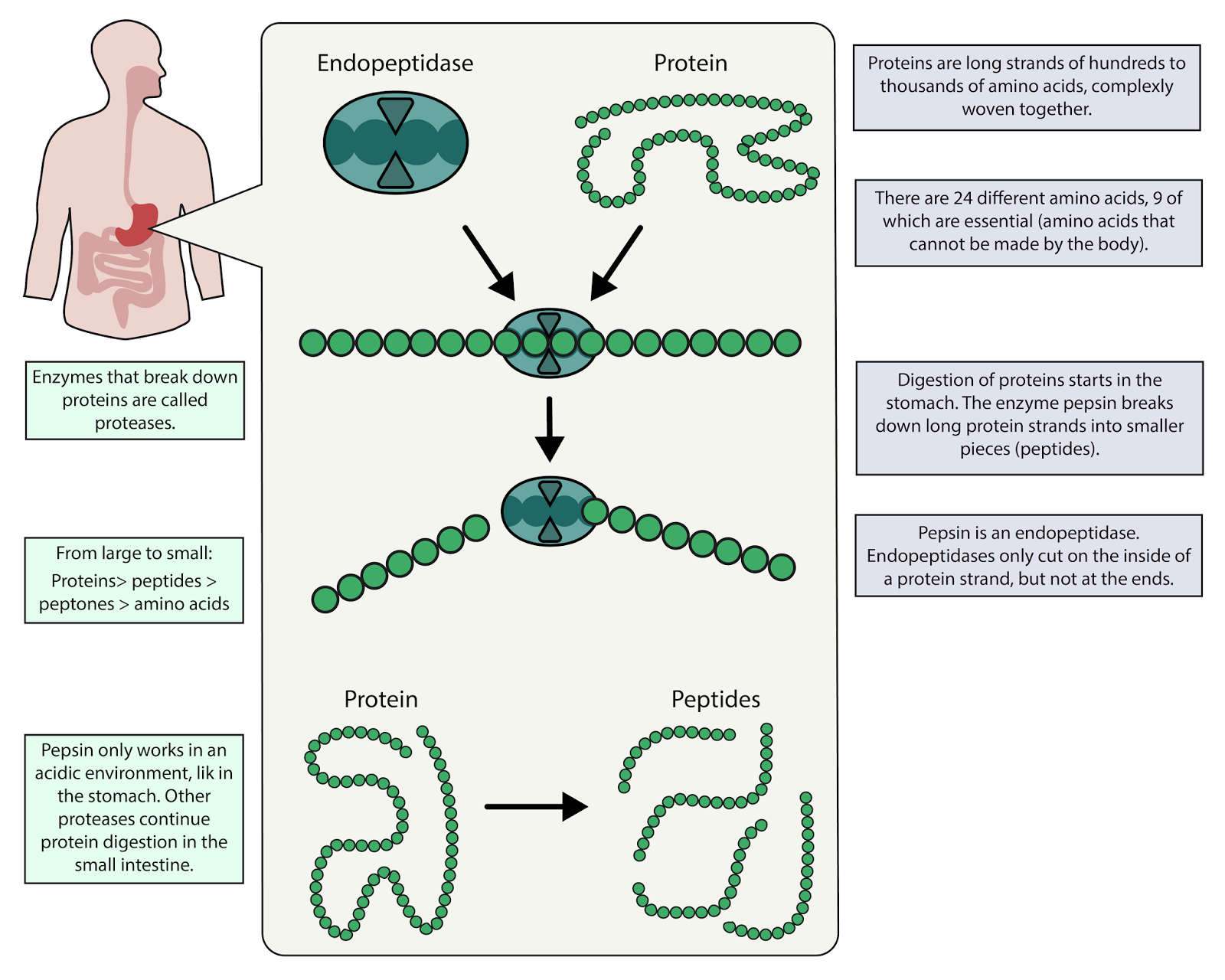
The latter is an enzyme that catalyzes the hydrolysis of peptide linkages at the free carboxyl end of the peptide chain, resulting in the stepwise liberation of free amino acids from the carboxyl end of the polypeptide.Proteases are enzymes in your digestive system that help break down food, acting like molecular-sized scissors that cut up proteins. Pancreatic juice also contains procarboxypeptidase, which is cleaved by trypsin to carboxypeptidase. Trypsin attacks peptide bonds involving the carboxyl groups of the basic amino acids (lysine and arginine). Chymotrypsin preferentially attacks peptide bonds involving the carboxyl groups of the aromatic amino acids (phenylalanine, tryptophan, and tyrosine). Both of these active enzymes catalyze the hydrolysis of peptide bonds in protein chains. They are activated in the small intestine as follows ( Figure 20.7 "Activation of Some Pancreatic Enzymes in the Small Intestine"): The intestinal mucosal cells secrete the proteolytic enzyme enteropeptidase, which converts trypsinogen to trypsin trypsin then activates chymotrypsinogen to chymotrypsin (and also completes the activation of trypsinogen). Pancreatic juice, carried from the pancreas via the pancreatic duct, contains inactive enzymes such as trypsinogen and chymotrypsinogen. Protein digestion is completed in the small intestine. It has a fairly broad specificity but acts preferentially on linkages involving the aromatic amino acids tryptophan, tyrosine, and phenylalanine, as well as methionine and leucine. Pepsin catalyzes the hydrolysis of peptide linkages within protein molecules. When food enters the stomach after a period of fasting, pepsinogen is converted to its active form-pepsin-in a series of steps initiated by the drop in pH. The principal digestive component of gastric juice is pepsinogen, an inactive enzyme produced in cells located in the stomach wall. HCl helps to denature food proteins that is, it unfolds the protein molecules to expose their chains to more efficient enzyme action. The pH of freshly secreted gastric juice is about 1.0, but the contents of the stomach may raise the pH to between 1.5 and 2.5. The hydrochloric acid (HCl) in gastric juice is secreted by glands in the stomach lining. In this chapter, we will look at each stage of catabolism-as an overview and in detail.įigure 20.6 The Principal Events and Sites of Protein Digestion In stage II, these monomer units (or building blocks) are further broken down through different reaction pathways, one of which produces ATP, to form a common end product that can then be used in stage III to produce even more ATP. One part of stage I of catabolism is the breakdown of food molecules by hydrolysis reactions into the individual monomer units-which occurs in the mouth, stomach, and small intestine-and is referred to as digestion The breakdown of food molecules by hydrolysis reactions into the individual monomer units in the mouth, stomach, and small intestine. In stage I, carbohydrates, fats, and proteins are broken down into their individual monomer units: carbohydrates into simple sugars, fats into fatty acids and glycerol, and proteins into amino acids. 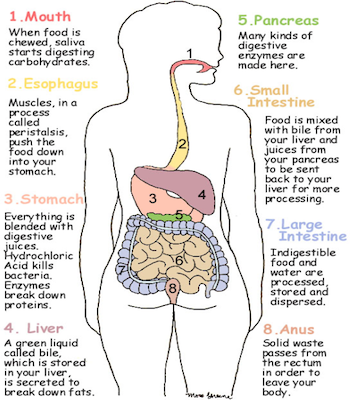
We can think of catabolism as occurring in three stages ( Figure 20.4 "Energy Conversions"). We have said that animals obtain chemical energy from the food-carbohydrates, fats, and proteins-they eat through reactions defined collectively as catabolism. Describe how carbohydrates, fats, and proteins are broken down during digestion. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
